Product Variants
Certified Quality
Support Available
Video laryngoscopy is increasingly used across Italian hospitals, operating theatres, and intensive care units as part of modern airway management practice. As clinical training and airway management approaches in Italy continue to align with European standards promoted by professional bodies such as SIAARTI, video assisted laryngoscopy plays an important role in improving visualization and first attempt intubation success.
AstraVue is a medical device manufacturer specializing in video laryngoscopy systems for airway management. The company designs and manufactures video laryngoscopes intended for routine and difficult airway management in operating rooms, intensive care units, and emergency settings.

AstraVue designs and manufactures video laryngoscopes and blade systems developed for clinical use in international healthcare markets. The product range is built to support reliable airway visualization while remaining simple and intuitive for clinicians working in high pressure environments.
AstraVue manufactures reusable, disposable, and hybrid video laryngoscope blade systems, allowing hospitals and clinics to select configurations that align with their clinical workflows and infection control requirements. All devices are developed in alignment with European medical device regulations for use in regulated healthcare systems, including Italy.

Italian anesthesia and intensive care practice places strong emphasis on patient safety and controlled airway management. Video laryngoscopy improves glottic visualization compared to direct laryngoscopy, helping clinicians better assess airway anatomy and reduce the risk of failed or repeated intubation attempts.
This is particularly important in operating rooms and emergency settings where rapid and accurate airway access is critical.
Video laryngoscopy supports higher first attempt intubation success in both routine and difficult airways. In Italian hospitals managing diverse patient populations, including critically ill and high risk cases, this improves clinical efficiency and reduces airway related complications.
Better first pass success also means less airway trauma and lower physiological stress for patients.
Shared screen visualization allows multiple clinicians to view the airway during intubation, supporting supervision, teaching, and team coordination. This aligns well with structured anesthesia and intensive care training practices used across Italy.
Video laryngoscopy helps junior clinicians learn under direct guidance while maintaining patient safety in real clinical environments.

Hybrid blade systems combine reusable components with disposable elements, offering flexibility between cost management and infection prevention strategies.

Disposable blades are designed for single use settings where infection control and rapid turnover are priorities, such as emergency departments and critical care units.
AstraVue video laryngoscopes are designed and manufactured to support predictable and consistent airway visualization across a wide range of clinical scenarios. The systems combine high resolution camera imaging for clear visualization of airway anatomy with medical grade construction suitable for frequent clinical use. Ergonomic handling supports controlled and precise intubation, while straightforward operation helps reduce cognitive load during airway management. The overall design philosophy focuses on clarity and reliability rather than unnecessary complexity.
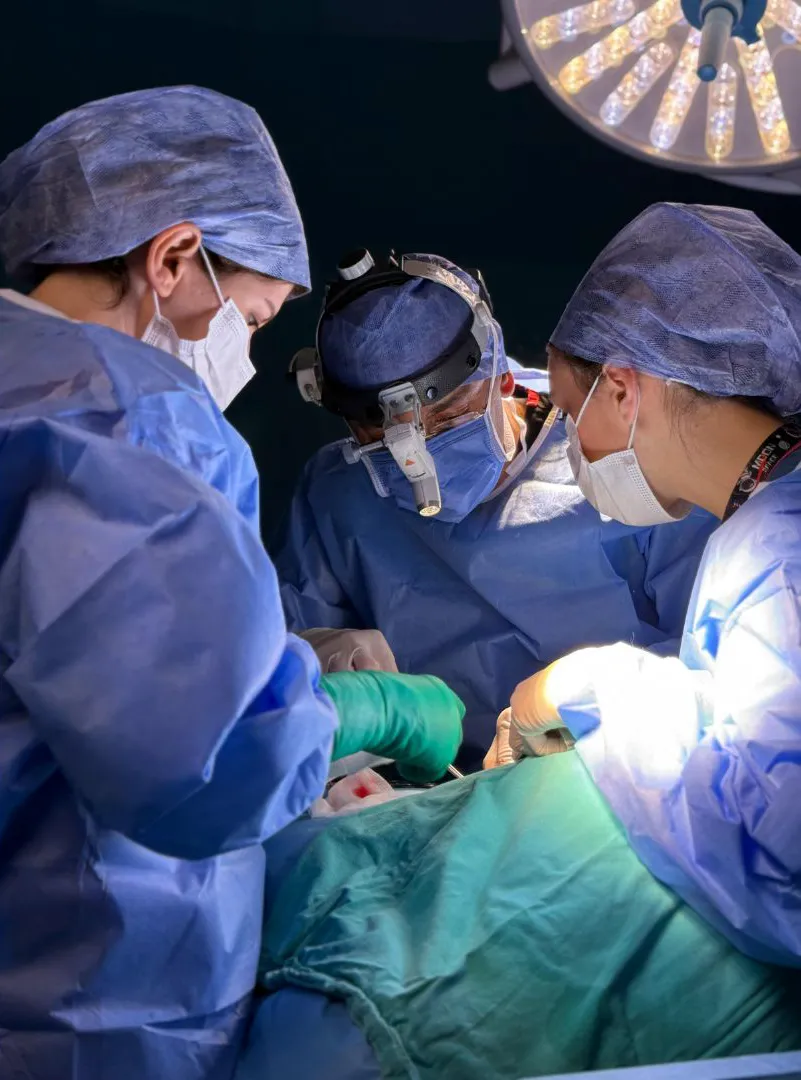
AstraVue video laryngoscopes are intended for use across a wide range of clinical environments, including:
The external display enables multiple clinicians to view the airway simultaneously, supporting team-based airway management and structured training approaches commonly used in Italian anesthesia and intensive care education.
Medical devices used in Italy must comply with European Union medical device regulations, which form the regulatory framework adopted nationally.
AstraVue video laryngoscopes are developed in alignment with EU medical device regulations and CE marking requirements applicable to devices placed on the Italian market under the oversight of the Italian Ministry of Health.
This ensures the devices meet established safety, performance, and quality system requirements for clinical use in Italy.
AstraVue supports structured engagement with healthcare providers and distribution partners in Italy.
Hospitals and clinics can request detailed technical specifications and clinical use information to support internal evaluation and procurement discussions.
Distributors interested in representing an export-quality medical device manufacturer can engage in discussions focused on long-term collaboration and regional market support.
If you are evaluating video laryngoscopy systems for clinical use in Italy or exploring distribution opportunities, you can request detailed product information, technical specifications, and usage guidance.
The AstraVue team supports informed clinical and commercial discussions without obligation.

Our global network of authorized distributors provides comprehensive support to healthcare professionals. For product inquiries, contact your local distributor. For distribution opportunities and partnership inquiries, contact us directly.
Yes. Medical devices used in Italy must comply with European Union medical device regulations. Devices placed on the Italian market must meet CE marking requirements and fall under the regulatory oversight of the Italian Ministry of Health.
AstraVue is in the process of building partnerships with authorized distributors in Italy. Through these partnerships, we will provide sales support, product training, and after-sales service.
Our products meet global standards including FDA, CE, MDR, and ISO 13485 certifications, ensuring compliance with international healthcare regulations.
If distributors have stock available, delivery can be made within 1 week. If stock is not available, lead times may extend up to 20 weeks (approximately 5 months)
The monitor provides 120 minutes of continuous operation and features quick recharge capability for uninterrupted clinical use.
Yes, comprehensive training support including hands-on instruction and educational materials is available through our authorized distributors to ensure optimal use of our systems.
Yes, the monitor features external mini HDMI connectivity, allowing you to connect to larger displays for training sessions, case reviews, or teaching demonstrations.
Our blade range includes pediatric sizes starting from Miller 000 and 00, as well as Macintosh sizes 1 and 2, ensuring appropriate options for infant and pediatric airway management.
Yes, we are actively expanding our global distribution network. We seek qualified partners with experience in medical device distribution, strong market presence, and commitment to clinical excellence. Contact us directly to discuss partnership opportunities in your territory.